Vol 7 No 4 2022- 18

Assessment of Thepax and Bio Boost for promoting microbial growth in common carp intestines Cyprinus carpio.
1 Aquaculture Unit, College of Agriculture, University of Basrah, Iraq; mohamedfawzi288@gmail.com
2 Aquaculture Unit, College of Agriculture, University of Basrah, Iraq; aaldubakel22@gmail.com.
Correspondence: aaldubakel22@gmail.com; Tel.: 00964780 350 4474
Available from: http://dx.doi.org/10.21931/RB/2022.07.04.18
ABSTRACT
The study aimed to evaluate the effect of Thepax and BioBoost as food additives on the microorganisms in the intestines of fish. From March 4 to October 22, the total number of common carp was 900 fish with an average weight of 163.41 ±10.16 g and a density of 100 fish/cage; three replicates were used for each treatment. The fish were fed three times daily. The included T1(0%additive), T2(1g/kgThepax)and T3 (1 g/kgBioBoost). The highest final weight value is Thepax treatment (2209.34 g), followed by Bio Boost and control. Microorganisms showed significant differences (P<0.05) in T2 for Lactobacillus sp. (10² x 65 CFU/ml) followed by T3 (10² x 55 CFU/ml) and control T1 (10² x 23 CFU/ml), also for Cellulomonas sp. in T2 (10² x 54 CFU/ml) followed by T3 (10² x 39 CFU/ml) and control T1 (10² x 7 CFU/ml). At the same time, Aeromonas sp. bacteria was higher in T1 (10² x 34 CFU/ml) over the treatments of T2 and T3 (10² x 2 CFU/ml) for both. We concluded the best additive was 1 g of Thepax / kg of feed in the recommended diets for common carp.
Keywords: Thepax; Additives; Microorganisms; Intestines; Bacteria; Lactobacillus; Endo Bio Boost
INTRODUCTION
Aquaculture is the fastest-growing food production and supports nearly 50% of all aquatic food for human consumption 1. The main objective of aquaculture is to increase production2. During cultivation, fish may be exposed to various diseases, especially if they are stressed due to the deterioration of water quality and exposure to stressful conditions, which leads to significant economic losses 3.
Antibiotics in aquaculture lead to many severe problems, such as antibiotic-resistant bacterial strains and antibiotic residues in fish muscles, which would cause serious public health effects on human consumers and environmental pollution 4,5. Therefore, finding safe and environmentally friendly alternatives is necessary to avoid the negative consequences of antibiotic use 6. Most non-nutritive feed additives include antibiotics, immunostimulants, antioxidants, probiotics and prebiotics 7,8, which were added to improve diet quality, health, performance and feeding efficiency of fish 9. Prebiotics are present in the cell wall of bacteria, yeasts and molds10,11. Prebiotics primarily affect the host by stimulating the selective growth of one or a limited number of non-pathogenic bacteria in the fish’s gut, resulting in improved host health. Lactic acid bacteria are the most important, as they are part of the natural microorganisms of the fish gut. All their strains are non-pathogenic, in addition to their antagonism against many types of pathogenic organisms through their production of lactic acid 12,13. The current study was conducted to evaluate the effect of Thepax and Bio Boost as feed additives on the microorganisms in the intestine of the common carp Cyprinus carpio.
MATERIALS AND METHODS
Study Site
The current study was accomplished in the ponds belonging to the Aquaculture Unit, which is located in the Agricultural Research and Experiment Station in Basrah Governorate (College of the Agriculture / University of Basrah), in Al-Hartha District, 16 km north of Basrah (300 65′ 64.6»N, 470 74′ 79.5»E). The current experiment was conducted in a large pond with 2500 m2. The water was provided with electric pumps from one branch of the Shatt Al-Arab river.
Fishes and Experimental Cages
Experimental cages were made of polyethylene with dimensions of length × width × height (3 × 1.7 × 1.8 m) and enclosed by external nets (10 × 10 mm). The cages were placed inside the earthen pond which was filled with water at the height of 1.5m; ventilation fans were employed to avoid the shortage of oxygen, especially during high temperatures. There were 600 common carp fish with an average weight of 163.41±10.16 g distributed in the cages at 100 fish/cage density; two replications for each treatment were used.
Feeding Management
Experimental diets were produced at the feed production plant of the Agricultural Advisory Office, Faculty of Agriculture, University of Basra. Three diets were formulated, which included a control T1 (0% additive), T2 (1g/kg Thepax) and T3 (1g/kg BioBoost). The pellet size was 6 mm. The ingredients used in the experimental diets are presented in Table (1), and the daily amount of feed for each cage was calculated based on body weight. Sampling was repeated every 20 days to monitor growth and adjust the amount of diet. The experiment lasted 233 days (from March 4 to October 22). The fis
h were fed daily, and the daily amount of feed for each treatment was divided into three equal meals; the first was in the morning (7 A.M.), the second was during noon, and the third was in the afternoon (4 P.M.).
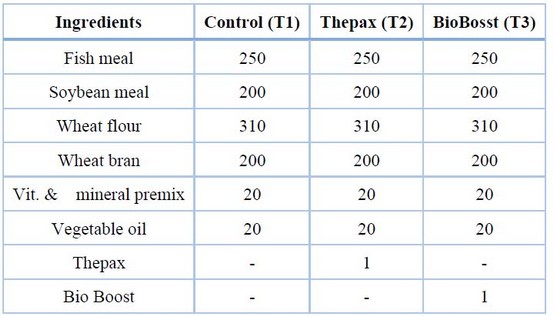
Table 1. Ingredients of the experimental diets (g/kg).
Environmental factors
Water’s most significant environmental features were measured during the study period on the days of sampling. All measurements were taken within nine A.M., including water temperature, pH, salinity, and dissolved oxygen.
Growth performance
Growth criteria were used to describe the growth performance of common carp fed the experimental diets as follows:
Weight gain:
WG =Final weight (g) – Initial weight (g)
Survival rate ( % ):
Survival rate = (No. of fish at the end / Total No. of fish) ×100
Hematology
Blood was drawn from different groups of fish before and after the experiment. After cutting the caudal peduncle, the fish were held vertically with the head up to help blood flow by pressing on the caudal vein. Capillaries and tubes containing an anticoagulant were used. They were transferred to the laboratory to test the hematocrit, red blood cells (RBC) and white blood cells (WBC), according to Witeska et al.(2022).
Gut Sampling
Before and after the experiment, three fish were randomly sampled from each treatment and transported alive to the laboratory. The intestines were isolated from the body cavity using sterile scissors and forceps, and the contents were collected into sterile 2 ml tubes by squeezing with forceps.
rinciple of the pour plate method
In this method, serial dilutions of the inoculum (serially diluting the primary specimen) are added within sterile Petri plates to which is poured melted and cooled (42-45°C) agar medium and completely mixed by revolving the plates, which are then left to solidify.
After incubation, the plates are observed for the appearance of individual colonies growing everywhere in the medium. The pure colonies of varying sizes, shapes and colors may be isolated/transported into test tube culture media to prepare pure cultures.
Tested bacteria
– Lactobacillus spp.
– Falvobacterium spp.
– Aeromonas spp.
– Cellulomonas spp.
Chemical analysis
The experimental diets were analyzed according to AOAC (1990), moisture, protein, lipid, ash and fiber were determined, while nitrogen-free extract was calculated according to New (1987) as follows:
%NFE=%DM-(% EE+%CP+%ASH+%CF)
Where:
NFE = Nitrogen-free extract
DM = Dry matter
EE = Ether extract or crude lipid
CP = Crude protein
CF = Crude fiber Statistical analysis
The experiment data were analyzed with a completely randomized design (CRD). The differences between the treatment means were tested by analysis of variance (ANOVA), and the significant differences were tested by LSD test at 0.05 probability level using SPSS program Ver. 26.
RESULTS
Environmental factors
Table 2 shows the values of environmental factors during the study period, the highest water temperature in July (30°C) and the lowest in March (21°C), while changes in pH were limited (7.5 -8.1), the concentration of dissolved oxygen ranged between 7.2 mg / l in March to 6.5 mg / l in July. Water salinity fluctuated from 3.13 PSU in March to 4.84 PSU in July.
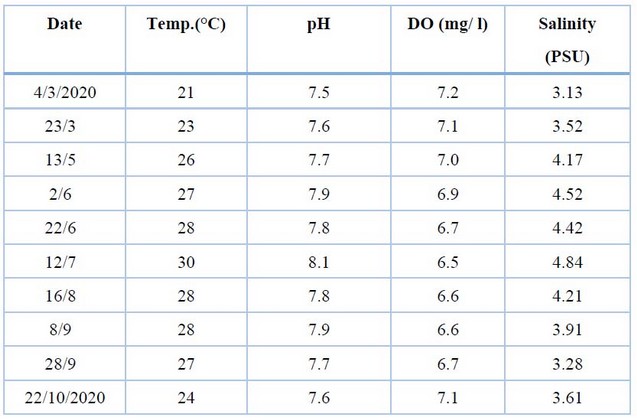
Table 2. The environmental factors during the study period.
Chemical analysis
The chemical composition of the experimental diet was presented in Table (3); the percentage of protein was 27.84% ±0.82, the lipid content was 6.39%±2.47, NFE was 44.20% ±2.35, and the Gross energy was 4272.50 Kcal.kg-1±60.10.
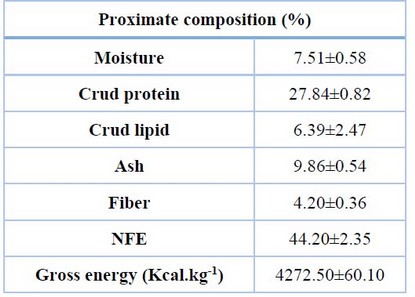
Table 3. Proximate composition of the experimental diet
Table (4) shows the averages of initial and final weight and the total weight gain of common carp for the different treatments. The highest value of the final weight was recorded in the Thepax treatment (2209.34 g), followed by Bio Boost (1918.27 g) and the lowest in the control treatment (1631.62 g). This was similar for weight gain, where the highest value was in Thepax (2035.65g), and the lowest value was recorded in the Control treatment (1478.23g).
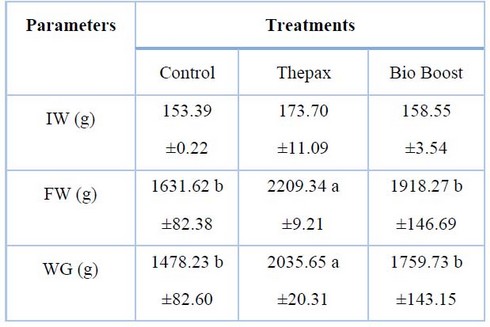
Table 4. Growth performances of common carp in different treatments
The blood parameters of common carp for different treatments showed in Table (5). It was noted that the hematocrit value did not differ significantly (P> 0.05) between the two treatments and the control. In contrast, all the hematocrit values for all the treatments were quite different (P<0.05) from the value of the pre-experiment fishes. Regarding the importance of red blood cells (RBC), it was significantly different (P<0.05) of the treatments of Thepax and BioBoost over the control treatment, which scored 1.543 and 1.533, respectively, then followed by the control treatments 1.257. While two treatments were significantly different (P<0.05) to the control treatment in the values of white blood cells (WBC) as Thepax and BioBoost treatments recorded 91.200 and 96.033, respectively, while the control treatment and pre-experiment recorded the least and amounted to 78.967 and 78.150 respectively.
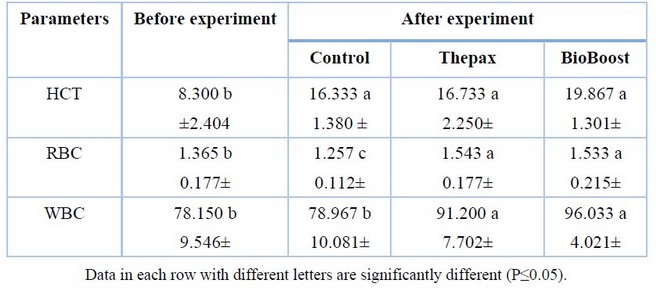
Table 5. Growth performances of common carp in different treatments
Gut microorganisms
Table (6) presented the numbers of bacteria in the intestines of common carp in different treatments. Where the number of colonies of Lactobacillus bacteria increased significantly (P<0.05) in the treatment of Thepax compared with the BioBoost treatment and control, as it recorded 10² x 65 cfu/ml and the BioBoost treatment recorded 10² x 55 cfu/ml followed by the control treatment which recorded 10² x 23 cfu/ml . In comparison, the highest rate of Aeromonas colonies was recorded in the intestines of fish in control, as it reached 10² x 34 CFU/ml. The colony numbers of these bacteria were significantly (P<0.05) reduced in the treatment of Thepax and BioBoost as it was recorded 10² x 2 CFU/ml for both treatments. The numbers of Flavobacterium increased significantly (P<0.05) in the control treatment, recording 10² x 33 CFU/ml. At the same time, it decreased in the Thepax and BioBoost therapy, as it recorded 10² X 3 CFU/ml and 10² X 7 CFU/ml, respectively. From Table (6), it was also noted that the number of colonies of cellulose-degrading bacteria Cellolomonas was significantly higher (P<0.05) in the Thepax treatment, recording 10² x 54 CFU/ml, followed by the BioBoost treatment with 10² X 39 CFU/ml, while the control treatment recorded 10² X 7 CFU/ml.
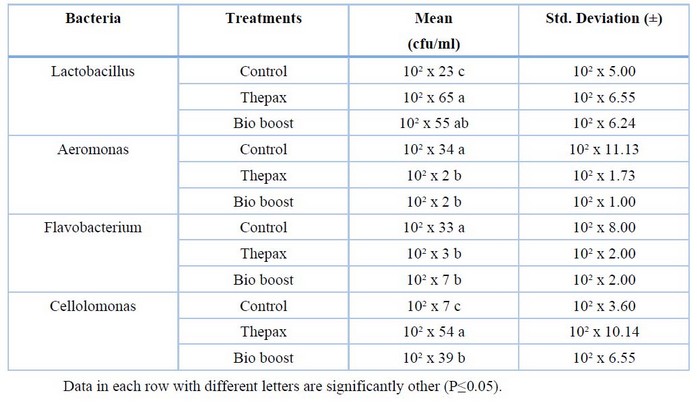
Table 6. Numbers of bacteria in the intestine of common carp in different treatments.
DISCUSSION
Water quality was considered the main factor for the success of fish culture projects 14; the water The temperature ranged from 21 to 30°C during the experiment, i.e., March to October. Hwang and Lin 15 found the optimum growth and FCR in common carp compared at 25°C. Song-bo et al. 16 observed the maximum daily feed intake at 28°C, which was in the range of the present study. The pH during the experimental period was within the appropriate limits for common carp culture, according to 16. The water salinity in the present study was within the applicable limits for common carp, which can tolerate a wide range of salinities17,18. Dissolved oxygen values in the entire study period were above 3 mg/l, and common carp optimal ranges between 4-8 mg/l 19,20.
The results obtained in the present study for growth parameters were in agreement with study 21, which found that the Thepax treatment was significantly higher than the control treatment. Al-Mhanawi et al.21 found that 1 g/kg Thepax has great potential as an important component for improving nutrient utilization and metabolism by increasing the surface area of the intestine in common carp and has a role in improving the digestive system compared to the control. The results were similar to those of study 22 by adding the prebiotic Mannan Oligosaccharide to trout feed, which showed a superior increase compared to other diets in terms of final weight. Al-Faragi 23 also increased weight indicator values by adding the prebiotic beta-glucan to common carp feed for eight weeks. While the results of the present study did not agree with those of Taher et al. 24 in grass carp (Ctenopharyngodon Idella) when adding 1 g/kg Thepax and 1 g/kg vitamin C to the diets, as no statistically significant differences were recorded with the control treatment in final weight and weight gain.
The survival rate of fish of the experimental therapies significantly increased compared to the control treatment. The best survival rate was recorded for the treatment of Thepax and BioBoost. A similar result was observed in the survival rate in the study of Tejpal et al.25on tilapia, where he confirmed that diets based on probiotics have a more positive effect on survival rate.
Blood parameters are considered one way to determine a fish’s health in terms of stress and disease26,27. The present study’s results agree with Akrami et al.,28, which revealed that common carp fed 1, 2, and 3 g/kg of the prebiotic MOS diet had significant superiority compared to the control in terms of hematocrit and white blood cells. The reason for this increase is due to the effect of the prebiotic on immune stimulation in fish. Modulation of the immune system is one of the generally proposed benefits of prebiotics and their ability to stimulate systemic immunity. Notably, several prebiotic supplements can even increase the rates of phagocytic cells, lysozyme cells and erythrocytes27.
The results of the current study indicate a clear superiority of the additive treatments over the control treatment in the intestinal content of colonies of beneficial bacteria Lactobacillus and Cellolomonas, especially the treatment of Thepax. These results also indicate a decrease in the number of colonies of pathogenic and harmful bacteria Aeromonas and Flavobacterium for the additive treatments compared to the control treatment. The results of the current study agree with Ringo28, in which probiotics were used in fish feed to enhance the immune response and increase fish resistance to diseases by increasing beneficial microorganisms in the intestine. Also, in the study of Yun Xia et al.29 in which probiotics from Bacillus cereus and Bacillus subtilis were used as food additives for tilapia, the results showed a stimulating effect on the group of beneficial microorganisms in the intestine. After stopping the intake of probiotics by the fish for one week, the beneficial bacteria in the intestine were very few. As for the results of Lv et al. 30 study, it was in agreement with the results of the current research, as FOS was used, which reduced Aeromonas bacteria in the intestines of hybrid Nile tilapia. And the study of Dimitroglou et al. 1 where nutritional supplements (MOS) were used, as a result, gave a significant reduction in the number of harmful bacteria Aeromonas in the intestines of rainbow trout.
CONCLUSIONS
The addition of Thepax enhanced growth performance, feeding efficiency, survival rate, increased Lactobacillus count and low Aeromonas bacteria in the intestines of common carp.
Author Contributions: A short paragraph specifying their individual contributions must be provided for research articles with several authors. The following statements should be used «Conceptualization, Jalal M. Al-Noor. and Adel Y. Al-Dubakel.; methodology, Mohamed F. Al-Janabi.; software, Jalal M. Al-Noor.; validation, Mohamed F. Al-Janabi, Jalal M. Al-Noor. and Adel Y. Al-Dubakel.; formal analysis, Adel Y. Al-Dubakel; investigation, Mohamed F. Al-Janabi; resources, Jalal M. Al-Noor; data curation, Adel Y. Al-Dubakel.; writing—original draft preparation, Mohamed F. Al-Janabi.; writing—review and editing, Jalal M. Al-Noor.; visualization, Adel Y. Al-Dubakel.; supervision, Adel Y. Al-Dubakel.; project administration, Jalal M. Al-Noor; All authors have read and agreed to the published version of the manuscript.
Funding: «This research received no external funding.»
Institutional Review Board Statement: «Not applicable.»
Informed Consent Statement: «Not applicable.»
Conflicts of Interest: «The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results».
REFERENCES
1. Dimitroglou A, Merrifield DL, Moate R, et al. Dietary mannan oligosaccharide supplementation modulates intestinal microbial ecology and improves gut morphology of rainbow trout, Oncorhynchus mykiss (Walbaum). J Anim Sci. 2009;87(10):3226-3234.
2. Abdel‐Latif HMR, Khafaga AF. Natural co‐infection of cultured Nile tilapia Oreochromis niloticus with Aeromonas hydrophila and Gyrodactylus cichlidarum experiencing high mortality during summer. Aquac Res. 2020;51(5):1880-1892.
3. Abdel‐Latif HMR, Dawood MAO, Menanteau‐Ledouble S, El‐Matbouli M. The nature and consequences of co‐infections in tilapia: A review. J Fish Dis. 2020;43(6):651-664.
4. Adel M, Dadar M, Oliveri Conti G. Antibiotics and malachite green residues in farmed rainbow trout (Oncorhynchus mykiss) from the Iranian markets: a risk assessment. Int J Food Prop. 2017;20(2):402-408.
5. Alderman DJ, Hastings TS. Antibiotic use in aquaculture: development of antibiotic resistance–potential for consumer health risks. Int J food Sci Technol. 1998;33(2):139-155.
6. Abdel‐Tawwab M, El‐Ashram AM, Tahoun A, Abdel‐Razek N, Awad SMM. Effects of dietary sweet basil (Ocimum basilicum) oil on the performance, antioxidants and immunity welfare, and resistance of Indian shrimp (Penaeus indicus) against Vibrio parahaemolyticus infection. Aquac Nutr. 2021;27(4):1244-1254.
7. Kord MI, Srour TM, Omar EA, Farag AA, Nour AAM, Khalil HS. The immunostimulatory effects of commercial feed additives on growth performance, non-specific immune response, antioxidants assay, and intestinal morphometry of Nile tilapia, Oreochromis niloticus. Front Physiol. 2021;12:627499.
8. Dawood MAO, Koshio S, Esteban MÁ. Beneficial roles of feed additives as immunostimulants in aquaculture: a review. Rev Aquac. 2018;10(4):950-974. doi:https://doi.org/10.1111/raq.12209
9. Al-Dubakel AY, Al-Hamadany QH, Mohamed AA. Effect of local probiotic (Iraqi probiotic) on the growth of common carp Cyprinus carpio L. youngs. J Basrah Res. 2015;41:57-69.
10. Hukins RW, Krumbeck JA, Bindels LB, et al. Prebiotics: why definition matter. Curr Opin Biotechnol. 2016;37:1-7.
11. Irianto A, Austin B. Probiotics in aquaculture J Fish Dis 25 (11): 633-642. doi. org. Published online 2002.
12. Ararame FG. Prebiotic effects on energy source preference, gut morphology and feed intake regulation in common carp (Cyprinus carpio carpio) during mild heat stress. Published online 2011.
13. Najim, Y. S., Mohammed, Th. T. & Al-Khalani, F. M. H. The effect of the use of different levels of Azolla to male broilers diets in the production and physiological performance and economic feasibility. Biochemical and Cellular Archives. 2020, 20(1): 573-580. Doi: 10.35124/bca.2020.20.1.573
14. Björnsson B, Ólafsdóttir SR. Effects of water quality and stocking density on growth performance of juvenile cod (Gadus morhua L.). ICES J Mar Sci. 2006;63(2):326-334.
15. Hwang DF, Lin TK. Effect of temperature on dietary vitamin C requirement and lipid in common carp. Comp Biochem Physiol Part B Biochem Mol Biol. 2002;131(1):1-7.
16. Song-bo C, Wei-xing C, Zhao-ting F. Effect of water temperature on feeding rhythm in common carp (Cyprinus carpio haematopterus Temminck et Schlegel). J Northeast Agric Univ (English Ed. 2012;19(1):57-61.
17. Al-dubakel AY, Al-noor SS, Mojer AM, Taher MM, Muhammed SJ. Comparison of Growth of Common Carp , Cyprinus carpio Cultivated in and outside Cages in Earthen Ponds in Basrah , Iraq. 2022;6(2):204-217.
18. Salati AP, Baghbanzadeh A, Soltani M, Peyghan R, Riazi G. Effect of different levels of salinity on gill and kidney function in common carp Cyprinus carpio (Pisces: Cyprinidae). Ital J Zool. 2011;78(3):298-303. doi:10.1080/11250003.2011.567400
19. Bauer C, Schlott G. Reaction of common carp (Cyprinus carpio, L.) to oxygen deficiency in winter as an example for the suitability of radio telemetry for monitoring the reaction of fish to stress factors in pond aquaculture. Aquac Res. 2006;37(3):248-254.
20. Linhart O, Rodina M, Gela D, Kocour M, Rodriguez M. Improvement of common carp artificial reproduction using enzyme for elimination of egg stickiness. Aquat Living Resour. 2003;16(5):450-456.
21. Al-Jubouri BS, Saleh KI. Effect of using bacterial-enzyme mixture, bread yeast and Thepax on production performance of common carp (Cyprinus carpio L.) cultured in floating cages. J Univ Babylon. 2017;25(4):1406-1414.
22. Denji KA, Mansour MR, Akrami R, Ghobadi S, Jafarpour SA, Mirbeygi SK. Effect of dietary prebiotic mannan oligosaccharide (MOS) on growth performance, intestinal microflora, body composition, haematological and blood serum biochemical parameters of rainbow trout (Oncorhynchus mykiss) juveniles. J Fish Aquat Sci. 2015;10(4):255.
23. Al-Faragi JK. The Efficacy of Prebiotic ([Beta]-Glucan) as a Feed Additive against Toxicity of Aflatoxin B1 in Common Carp, Cyprinus Carpio L. J Aquac Res Dev. 2014;5(4):1.
24. Taher MM, Muhammed SJ, Mojer AM, Al-Dubakel AY. The Effect of Some Food Additives on Growth Parameters of Grass Carp Ctenopharyngodon idella Fingerlings. Basrah J Agric Sci. 2022;35(1):120-131.
25. Dahiya T, Sihag RC, Gahlawat SK. Effect of probiotics on the haematological parameters of Indian Magur (Clarius batrachus L.). J Fish Aquat Sci. 2012;7(4):279.
26. Fazio F, Marafioti S, Torre A, Sanfilippo M, Panzera M, Faggio C. Haematological and serum protein profiles of Mugil cephalus: effect of two different habitats. Ichthyol Res. 2013;60(1):36-42.
27. Gelibolu S, Yanar Y, Genc MA, Genc E. The effect of mannan-oligosaccharide (MOS) as a feed supplement on growth and some blood parameters of Gilthead Sea Bream (Sparus aurata). Turkish J Fish Aquat Sci. 2018;18(6):817-823.
28. Akrami R, Chitsaz H, Hezarjaribi A, Ziaei R. Effect of dietary mannan oligosaccharide (MOS) on growth performance and immune response of Gibel carp juveniles (Carassius auratus gibelio). J Vet Adv. 2012;2(10):507-513.
29. Xia Y, Wang M, Gao F, Lu M, Chen G. Effects of dietary probiotic supplementation on the growth, gut health and disease resistance of juvenile Nile tilapia (Oreochromis niloticus). Anim Nutr. 2020;6(1):69-79.
30. Zhou Z, Ding Z, Huiyuan L V. Effects of dietary short‐chain fructooligosaccharides on intestinal microflora, survival, and growth performance of juvenile white shrimp, Litopenaeus vannamei. J World Aquac Soc. 2007;38(2):296-301.
Received: July 20 2022 / Accepted: October 15 2022 / Published:15 November 2022
Citation: Al-Janabi M, Al-Noor J, Al-Dubakel A Y. Assessment of Thepax and Bio Boost for promoting microbial growth in common carp intestines Cyprinus carpio. Revis Bionatura 2022;7(4) 18. http://dx.doi.org/10.21931/RB/2022.07.04.18




















